Edentulous maxilla: First global consensus for treatment

Key Takeaways for Busy Clinicians
- There is no single “best” approach—case selection determines success
- Grafting vs. graftless is a strategic decision, not a preference
- The new global consensus provides a practical framework for these decisions

Read the full first Global Consensus publication here.
Why This Consensus Is Important
The first Global Consensus for Clinical Guidelines was developed in response to ongoing clinical and patient‑centered challenges associated with rehabilitating the edentulous maxilla.
The wide variability in anatomy and available treatment approaches has highlighted the need for clear, harmonized recommendations. Furthermore, the initiative also addresses the inherent limitations of regional consensus efforts. Through broad international participation, with patient input integrated alongside clinician expertise, the initiative resulted in a unified global guideline.
Interview with Prof. Frank Schwarz
Want expert insights behind the consensus? In our interview, Prof. Schwarz explains what these guidelines mean for daily clinical practice.
Methodology
The guideline follows an S2k‑level methodological framework and a rigorous, transparent, and well‑structured process. It brings together evidence from eight systematic reviews, six structured surveys, and a three‑round Delphi procedure, supported by contributions from 105 delegates from 26 countries. The outcome is the first global collection of 36 consensus‑based recommendations, each linked to a newly defined Core Outcome Set for treating the edentulous maxilla.
Key Outcomes
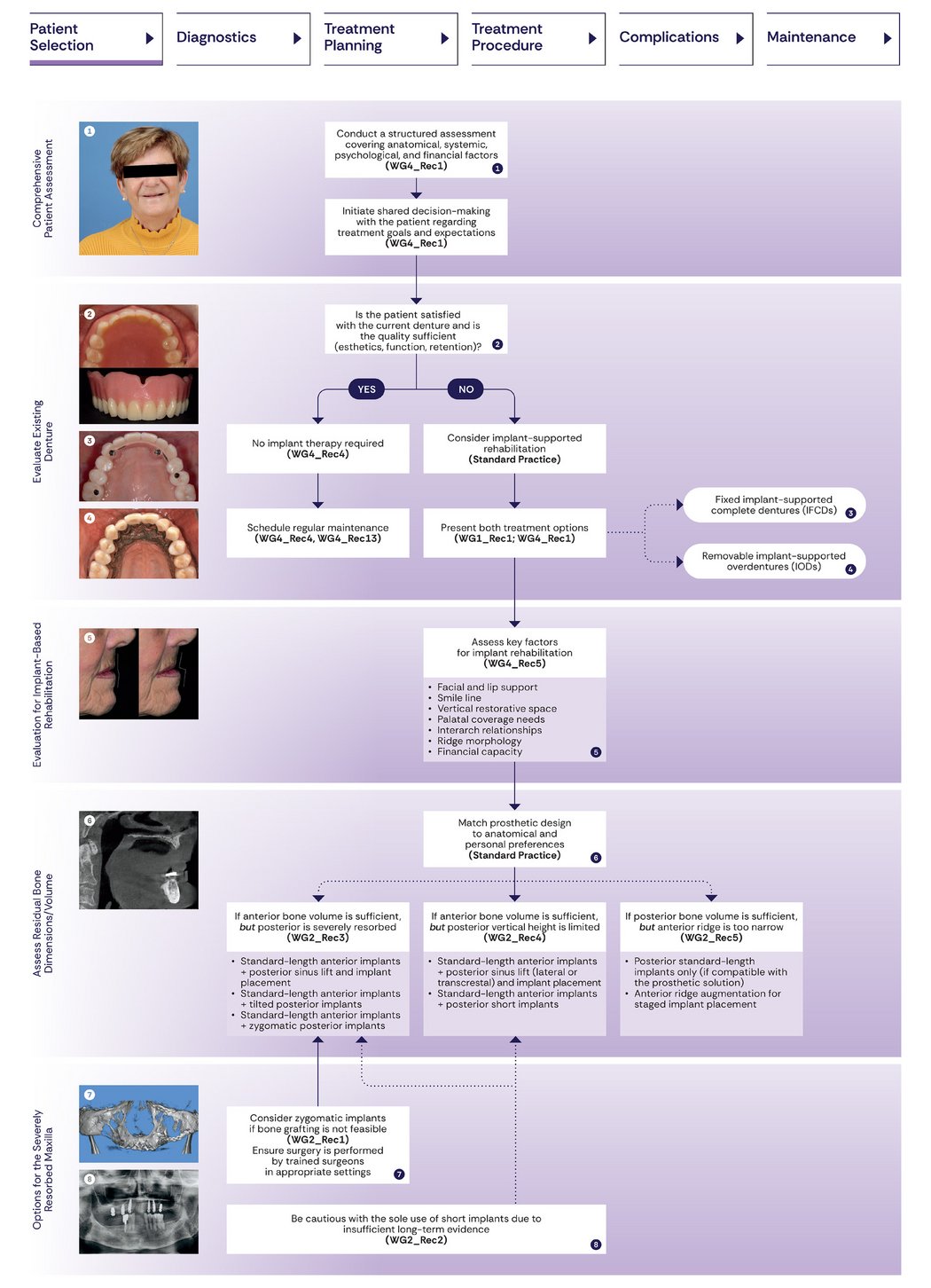
1. A Fully Structured Clinical Workflow
A major outcome of the consensus is a stepwise, structured workflow. It covers patient selection, diagnostics and prosthetic planning, surgical and prosthetic treatment, management of complications, and long-term maintenance.
This structured approach provides clinicians with a predictable, evidence‑based pathway from initial assessment through long‑term care, supporting consistent practice across different regions and clinical settings.
In addition, newly developed decision trees and checklists facilitate the implementation of the recommendations in daily clinical practice.
2. A Shift Toward PatientCentered Care
Traditionally, outcomes of implant therapy with fixed full‑arch prostheses have relied on objective clinician‑reported outcomes such as survival rates and marginal bone levels, which do not always reflect what matters most to patients. Recent research has therefore increasingly emphasized the importance of patient‑reported outcomes (PROs). The consensus addresses this gap by formally embedding PROs into every stage of care. Within the Core Outcome Set, patient‑critical domains include esthetic satisfaction, functional comfort, overall treatment satisfaction, speech and phonetics, and oral health–related quality of life.
The consensus emphasizes shared decision-making by comparing conventional dentures, implant overdentures, and fixed prostheses. Patients must be fully informed about all available treatment options before therapy selection, reflecting a clear shift from clinician-driven care toward patient-informed, preference-based rehabilitation.
Read how Prof. Frank Schwarz explains the growing importance of patient-reported outcomes in regenerative success.
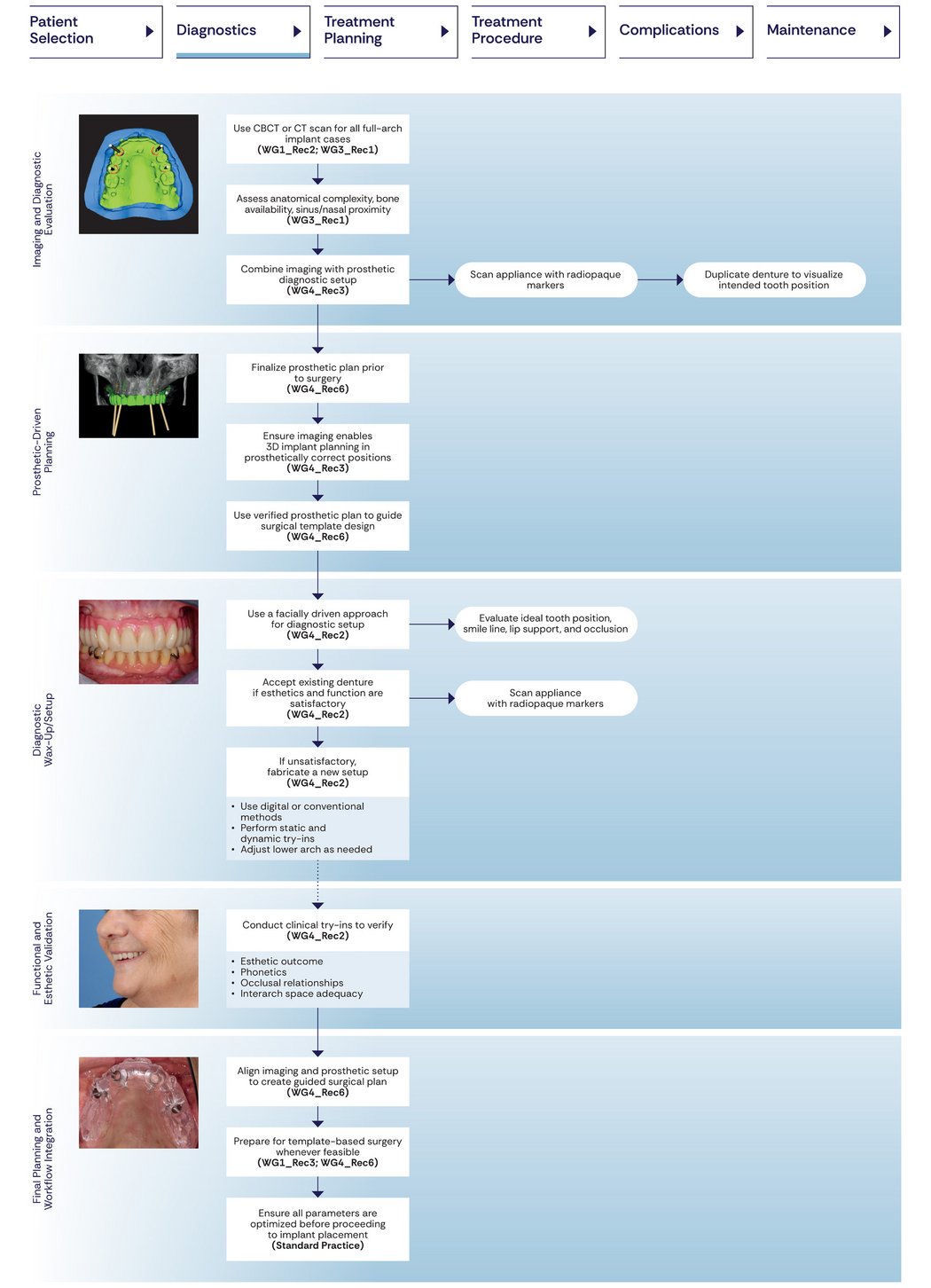
3. Diagnostics: CBCT and Prosthetically Driven Planning as the Standard of Care
One of the strongest cross‑group recommendations supports routine use of three-dimensional (3D) imaging, preferably cone beam computed tomography (CBCT), for all patients undergoing full-arch implant therapy, to evaluate maxillary anatomy, detect sinus conditions, plan augmentation procedures, and enable prosthetically driven implant placement.
The consensus places strong emphasis on prosthetically driven planning, highlighting that implant planning should be guided by the intended prosthetic outcome rather than surgical considerations alone. Whenever possible, the imaging process should be combined with a prosthetic diagnostic setup. By linking CBCT data with the planned prosthetic design, implant position and orientation can be aligned with the requirements of the final restoration, supporting functional, esthetic, and predictable treatment outcomes.
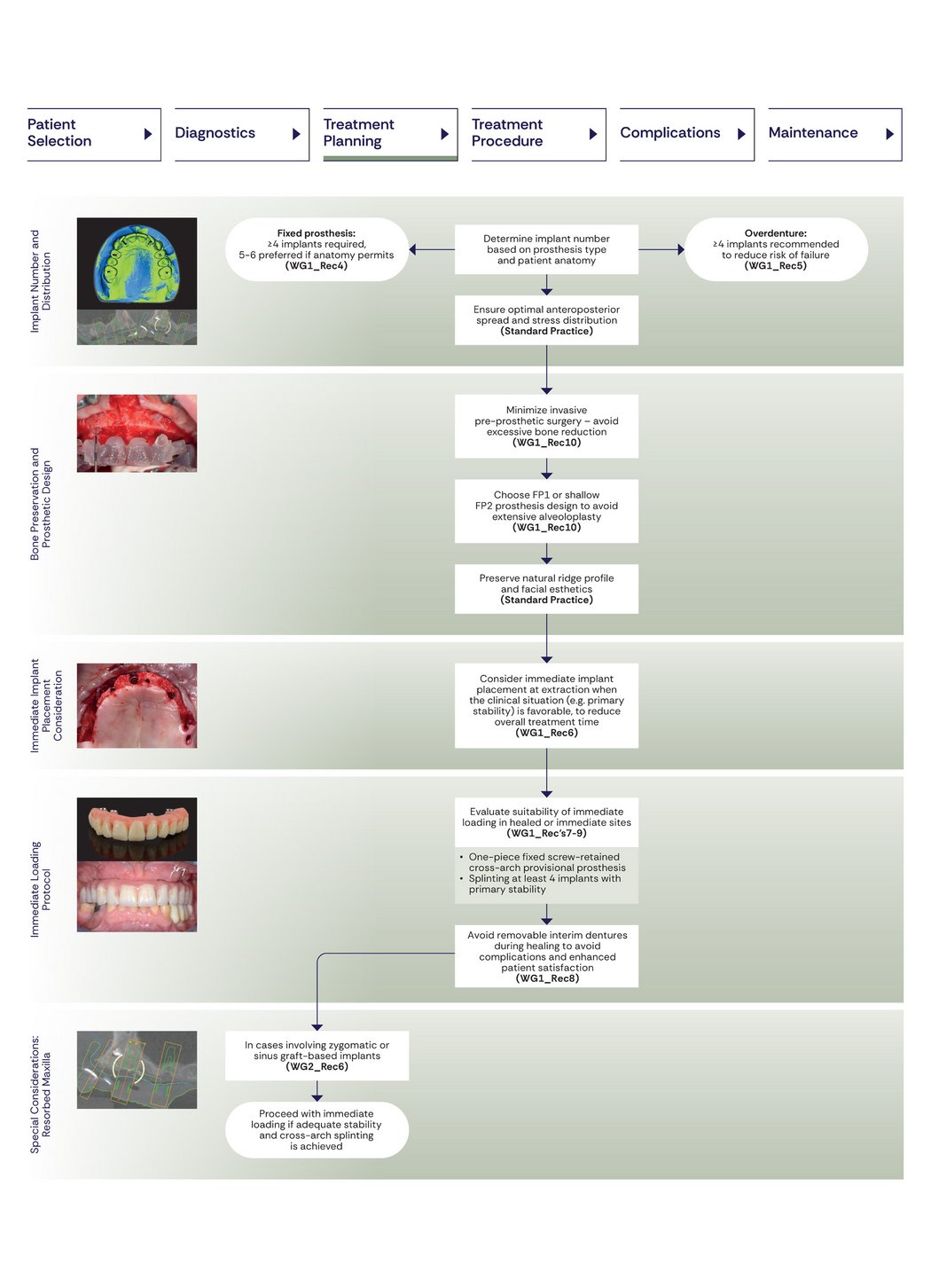
4. Treatment Planning: Defining Implant Numbers and Loading Protocols
Minimum Implant Numbers
The consensus offers clear guidance on how many implants to use and how they should be positioned. Decisions should be guided by the planned prosthetic solution, the patient’s anatomical conditions, and biomechanical considerations.
For fixed full‑arch prostheses, four implants represent the minimum requirement, while the placement of five or six implants is favored when anatomy permits, as this enhances stability and helps limit mechanical overload and technical complications. Implants should be positioned to optimize antero‑posterior spread and connected by a rigid cross‑arch framework, ideally fabricated as a single piece.
Timing of Placement and Loading
Immediate protocols, including immediate implant placement and immediate loading with a screw‑retained provisional full‑arch prosthesis, should be considered when clinical conditions are favorable and adequate primary stability can be achieved.
This aligns modern protocols with predictable success rates while reducing treatment time and enhancing patient experience.
5. Treatment Strategies Based on Bone Availability
The consensus provides structured guidance for treatment planning in patients with an edentulous maxilla based on available bone conditions.
When both anterior and posterior bone availability are limited, zygomatic implants may be considered a last‑line option and should be performed by highly experienced clinicians in appropriately equipped settings.
When sufficient anterior bone is present, but posterior bone height is inadequate, several treatment approaches may be considered, including the use of standard‑length implants combined with sinus floor elevation or other augmentation procedures, the placement of tilted posterior implants, or the use of zygomatic implants, acknowledging limitations mentioned above.
In this context, acceptance of hard‑tissue augmentation was high, with 80.5% of patients and 85.7% of cross‑disciplinary experts indicating they would be open to undergoing hard tissue augmentation procedures for a fixed prosthesis.
When posterior bone is available, but anterior ridge width is insufficient, treatment may involve placement of standard‑length posterior implants in combination with grafting procedures in the anterior region.
Overall, these recommendations form a clear, anatomy‑driven decision framework that can be adapted to individual clinical presentations.
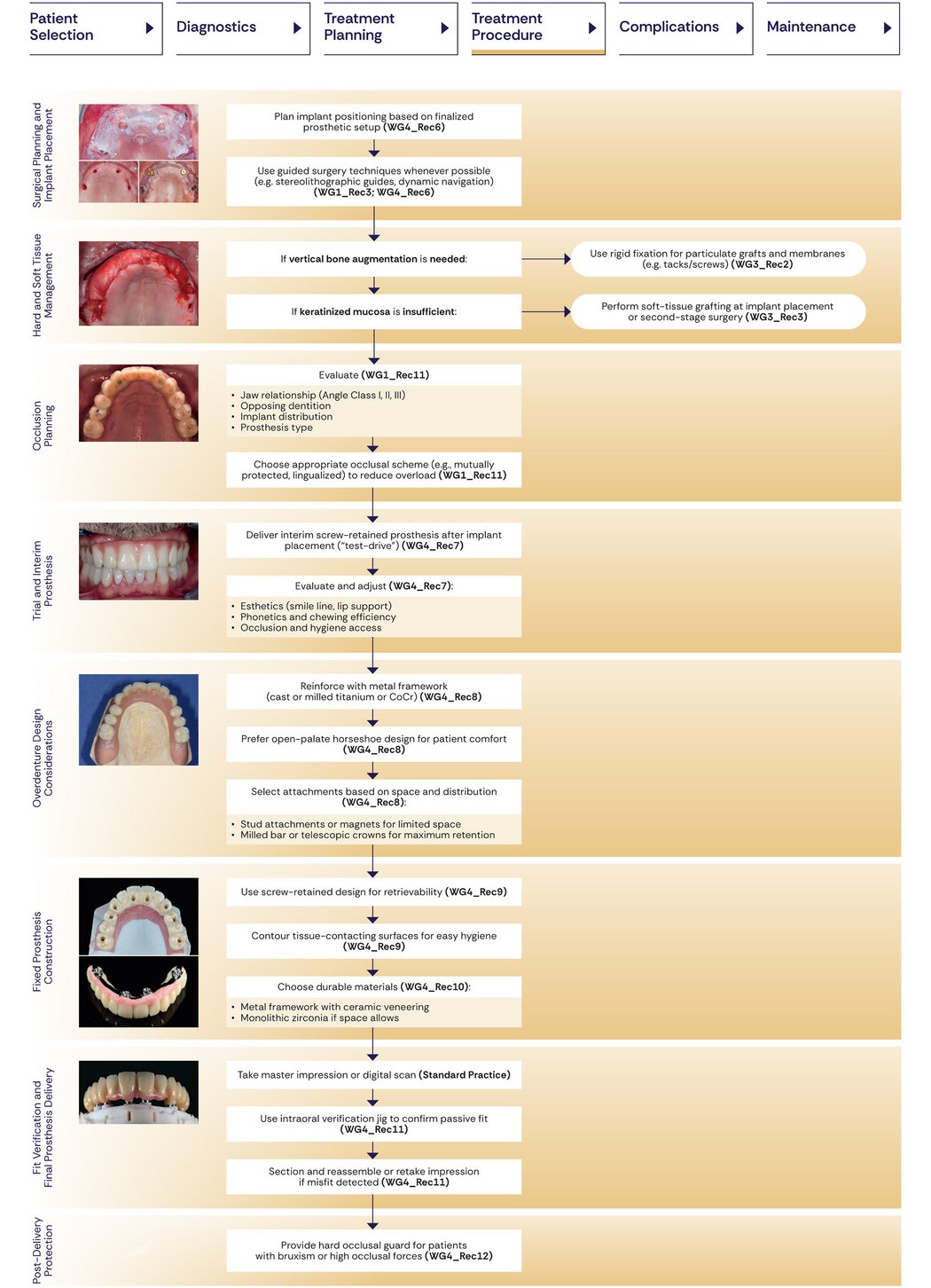
6. Hard-and Soft-Tissue Management
Consensus includes important high‑level recommendations on hard‑ and soft‑tissue management, both of which are considered essential for long‑term treatment success.
For hard‑tissue augmentation, in cases requiring vertical bone augmentation (e.g., onlay bone grafts or guided bone regeneration), particulate grafts and barrier membranes should be rigidly fixed with tacks or screws to prevent micro-movement. Early exposure of titanium meshes or non‑resorbable membranes during healing is associated with poorer outcomes and should be avoided.
For soft‑tissue augmentation, a consensus was reached on the need for grafting in cases of absent or thin keratinized mucosa, insufficient width of keratinized mucosa (< 2 mm), or lack of vestibular depth. Grafting may be performed at implant placement or during second‑stage surgery. Implant position and prosthesis type were not considered decisive factors.
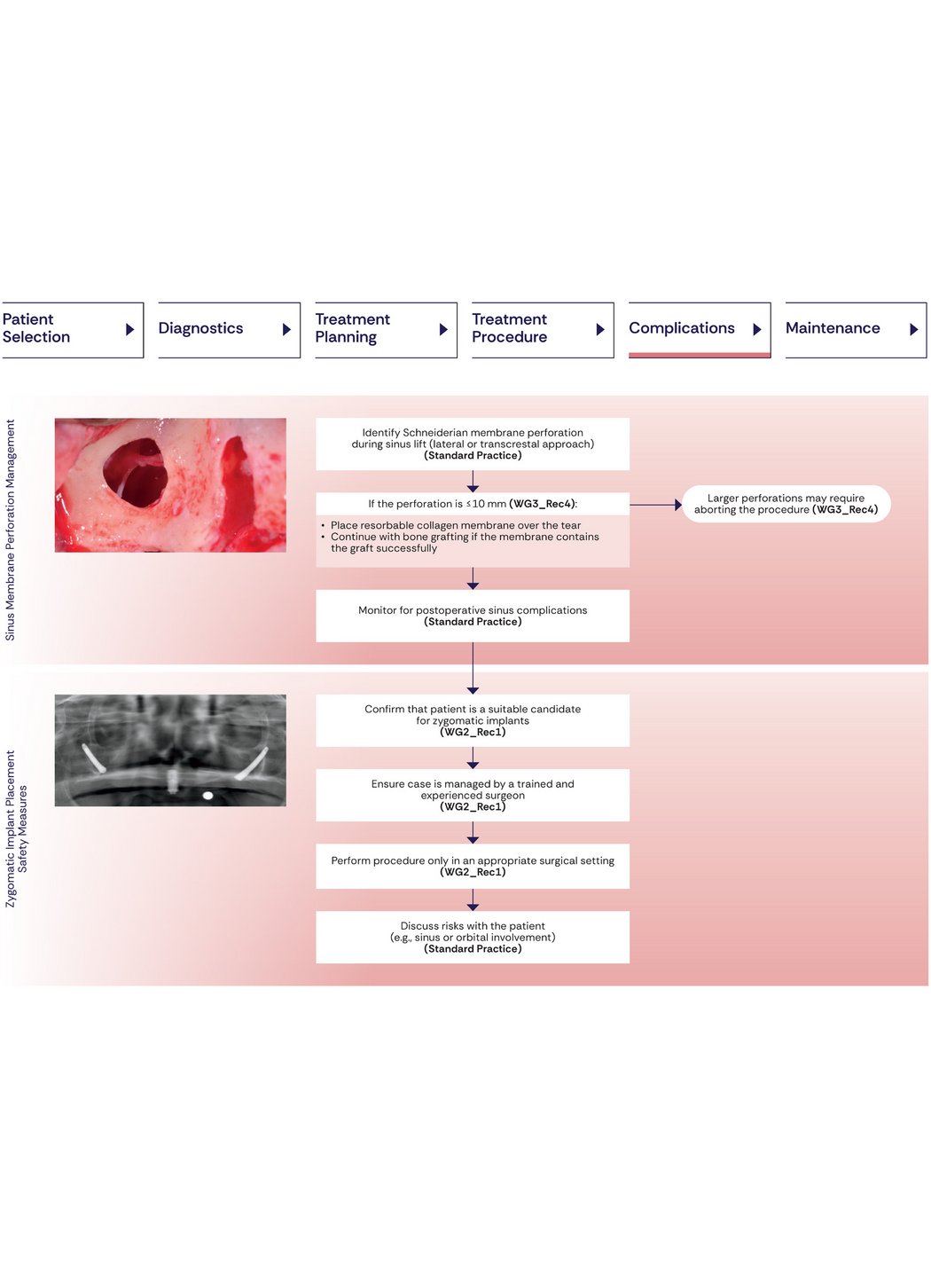
7. Management of Complications
An important contribution of the consensus is the provision of clear guidance for managing surgical complications, particularly Schneiderian membrane perforations during lateral sinus lift procedures. Management strategies depend on the size of the perforation. In cases of perforations measuring up to 5 mm, continuation of surgery is generally supported, with the perforation covered using a resorbable membrane. For perforations between 5 and 10 mm, careful placement of a resorbable collagen membrane over the tear is recommended to seal the defect before proceeding with the bone graft, provided the membrane can adequately contain the graft material. In cases of perforations greater than 10 mm, the use of bone blocks is not considered appropriate. Instead, delaying the augmentation or adopting a staged sinus lift is advised.
In addition, the consensus emphasizes careful case selection and the involvement of experienced surgical teams for complex interventions (zygomatic implant placement), to minimize complications and optimize outcomes.
8. The Role of Long‑Term Maintenance in Treatment Success
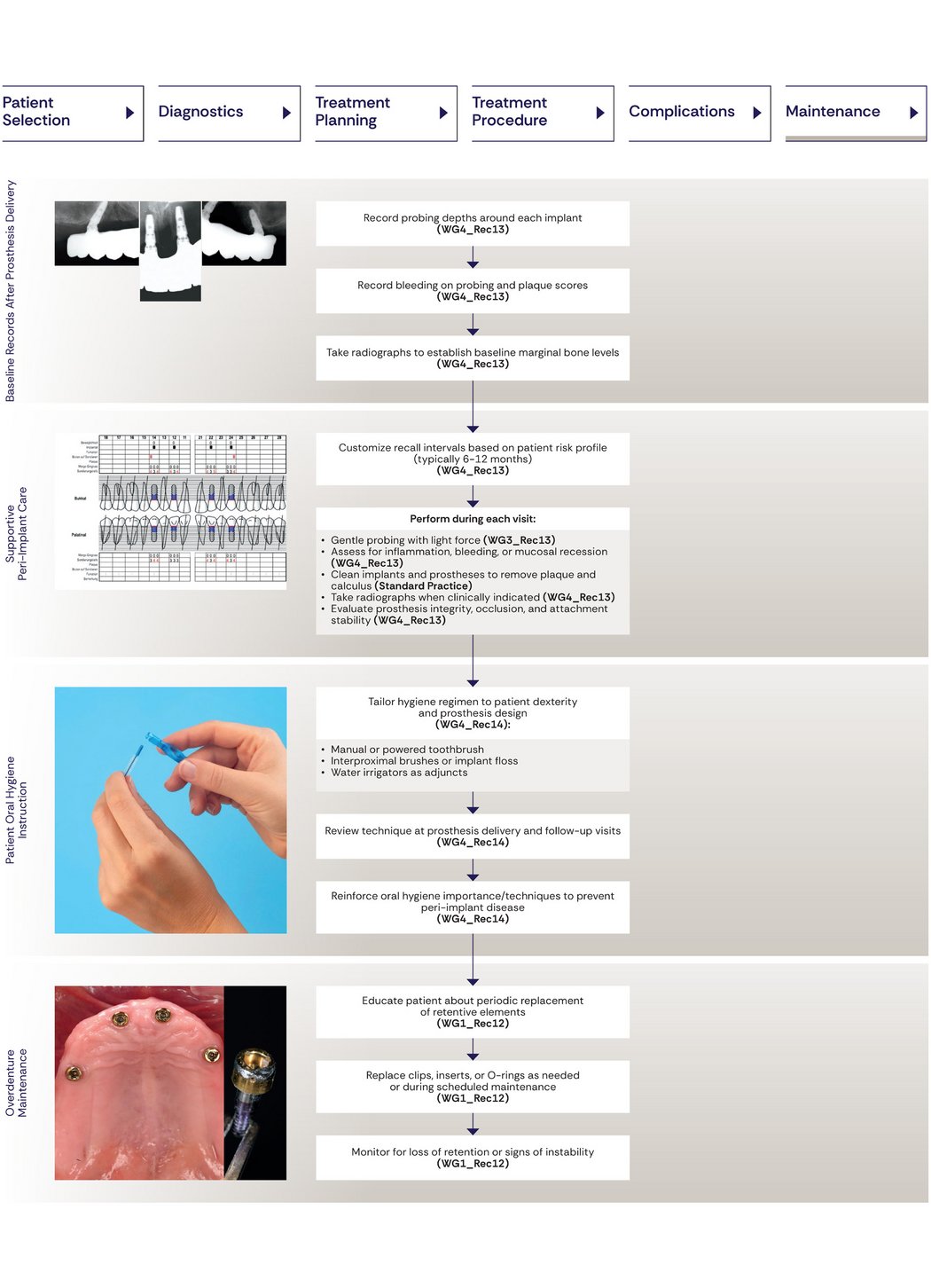
The consensus emphasizes that long‑term success in maxillary implant rehabilitation depends on structured maintenance and patient adherence. At the delivery of the final prosthesis, baseline peri‑implant parameters should be recorded, including probing depths, bleeding on probing, plaque indices, and intraoral radiographs to establish initial peri-implant marginal bone levels.
Follow‑up should be risk‑based, typically every 6–12 months, and include gentle peri‑implant probing, monitoring for bleeding or suppuration, assessment of soft‑tissue health, professional debridement, and radiographs when indicated to enable early detection of peri‑implant disease. Prosthesis design should allow adequate diagnosis and hygiene access.
Effective home care is essential and should be supported through individualized oral‑hygiene instruction and regular reinforcement. For implant‑supported overdentures, ongoing maintenance, including timely replacement of retention components, is required to maintain function and prevent complications.
9. Evidence Gaps and Areas for Future Research
Despite comprehensive recommendations, the consensus identifies several important evidence gaps. Further research is needed on the use of short implants for full‑arch rehabilitation, immediate loading protocols for short and zygomatic implants, standardized maintenance strategies, optimal recall intervals, and the diagnostic value of facial scanning. Additional work is also required to develop standardized patient‑reported outcome measures specific to the edentulous maxilla.
Conclusion: A New Era of Harmonized, Patient‑Centered Standards for Edentulous Maxilla Rehabilitation
The first Global Consensus marks an important milestone in implant dentistry by delivering a globally aligned, patient‑centered, and evidence‑based guideline for rehabilitation of the edentulous maxilla. It formalizes the full clinical workflow, places greater emphasis on patient priorities, clarifies surgical and prosthetic principles, and underscores the importance of maintenance and long‑term care. For clinicians and the broader healthcare community, this consensus offers a clear framework to support predictable, high‑quality implant care across diverse clinical settings worldwide.
Disclaimer
This article summarizes key findings and recommendations from the first Global Consensus on rehabilitation of the edentulous maxilla, as interpreted by Geistlich. Due to the extensive scope of the consensus, including multiple systematic reviews and surveys, not all recommendations could be included, and some details may have been unintentionally omitted. Errors cannot be entirely excluded. The authors of the consensus document are not responsible for the content of this summary.
References:
- Alcoforado G, et al. Clin Oral Impl Res, 2026, 37: S5-S6.
- Schwarz F, et al. Clin Oral Implants Res. 2026 Feb;37: S7-S27.
- Donos N, et al. Clin Oral Implants Res. 2026 Feb;37: S28-S48.
- Pala K, et al. Clin Oral Implants Res. 2026 Feb;37: S49-S67.
- Fiorellini J, et al. Clin Oral Implants Res. 2026 Feb;37: S68-S80.
- Stilwell C, et al. Clin Oral Implants Res. 2026 Feb;37: S81-S107.
- Brunello G, et al. Clin Oral Implants Res. 2026 Feb;37: S108-S120.
- Lin G-H, et al. Clin Oral Implants Res. 2026 Feb;37: S121-S134.
- Strauss FJ, et al. Clin Oral Implants Res. 2026 Feb;37: S135-S154.
- Schoenbaum TR, et al. Clin Oral Implants Res. 2026 Feb;37: S155-S165.
- Brunello G, et al. Clin Oral Implants Res. 2026 Feb;37: S166-S187
- Lin G-H, et al. Clin Oral Implants Res. 2026 Feb;37: S188-203
- Lin G-H, et al. Clin Oral Implants Res. 2026 Feb;37: S204-219
- Park J-Y, et al. Clin Oral Implants Res. 2026 Feb;37: S220-246
- Thoma DS, et al. Clin Oral Implants Res. 2026 Feb;37: S247-272
- Francisco H, et al. Clin Oral Implants Res. 2026 Feb;37: S273-301
- Romito GA, et al. Clin Oral Implants Res. 2026 Feb;37: S302-331
- Pannuti CM, et al. Clin Oral Implants Res. 2026 Feb;37: S332-345
- Sabri H, et al. Clin Oral Implants Res. 2026 Feb;37: S346-371
- Saleh MHA, et al. Clin Oral Implants Res. 2026 Feb;37: S372-394
- Saleh MHA, et al. Clin Oral Implants Res. 2026 Feb;37: S395-401